Organocatalytic depolymerization of poly(ethylene terephthalate)
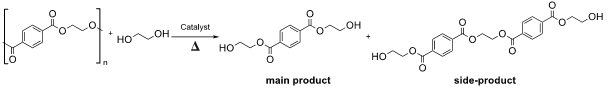
During my research work, I investigated the depolymerization of poly(ethylene terephthalate) (PET), because nowadays, our planet is facing climate change problems caused by pollution, and one of the main goals of humanity is to find the best way to diminish plastic waste and, eventually, recycle it and reuse it. Respectively, my aim was to perform the depolymerization of PET with the best reaction conditions. For my research work, I chose the glycolysis reaction (Figure 1) as a chemical recycling of PET. Multiple depolymerization reactions were conducted using several catalysts and different conditions to optimize the reaction.
The depolymerization reactions were carried out in sealed vials, under inert atmosphere (Ar), at a range of temperatures between 170 to 190 °C. Ethylene glycol was applied as reagent and solvent, and different Brønsted basic or H-bond donor organocatalysts supported on silica gel, such as the commercially available trialkyl amine, guanidine or thiourea functionalized silica gels, and the prepared 1,5,7-triazabicyclo[4.4.0]dec-5-ene (TBD) functionalized silica gel were used as catalysts. After obtaining the main product bis(hydroxyethyl)terephthalate (BHET) and side products, too, we characterized them by NMR and HPLC-MS techniques, and the stability of the abovementioned supported organocatalysts was determined by TG-DSC technique.
The optimization of the glycolysis reaction was mainly accomplished based on four aspects: the type of supported catalyst, the reaction temperature, the ratio of catalyst: PET, and ratio of solvent: PET. Finally, we found a method resulting in almost quantitative production (96%) of BHET, which is a readily appropriate starting material for preparing PET.
szerző
-
Daicu Diana
Vegyészmérnöki alapképzési szak, nappali BSC
alapképzés (BA/BSc)
konzulensek
-
Dr. Kupai József
egyetemi docens, Szerves Kémia és Technológia Tanszék -
Fehér Zsuzsanna
PhD hallgató, Szerves Kémia és Technológia Tanszék -
Kisszékelyi Péter
doktorjelölt, Szerves Kémia és Technológia Tanszék

